Quantification of the P=O?HN hydrogen bond in the binding of creatinine with phosphonate calix [4]pyrroles
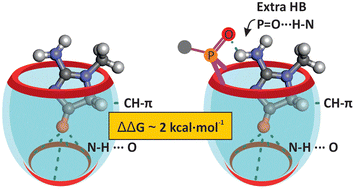
We report the results of 1H and 31P NMR titrations of a series of calix[4]pyrrole cavitands with hexyl creatinine in dichloromethane solution. We show that all studied calix[4]pyrrole cavitands form thermodynamically and kinetically highly stable 1 : 1 inclusion complexes. We used isothermal titration calorimetry (ITC) experiments to thermodynamically characterize the inclusion complexes. The binding processes are enthalpically and entropically favored. The cavitand receptors incorporating an inwardly directed phosphonate bridging group at their upper rim produce more stable complexes. The X-ray structure of the inclusion complex of a mono-phosphonate calix[4]pyrrole cavitand having an inwardly directed phosphonate group with the creatinine derivative reveals the existence of an intramolecular P![[double bond, length as m-dash]](https://iciq.org/wp-content/uploads/2024/09/char_e001.gif) O⋯HN hydrogen bond. We estimated that the strength of the P
O⋯HN hydrogen bond. We estimated that the strength of the P![[double bond, length as m-dash]](https://iciq.org/wp-content/uploads/2024/09/char_e001.gif) O⋯HN hydrogen bond is close to 2 kcal mol−1 based on the differences in free binding energies between complexes having the inwardly directed P
O⋯HN hydrogen bond is close to 2 kcal mol−1 based on the differences in free binding energies between complexes having the inwardly directed P![[double bond, length as m-dash]](https://iciq.org/wp-content/uploads/2024/09/char_e001.gif) O group and those lacking it. We performed DFT calculations to gain some insight into the structures of the inclusion complexes and support the experimental findings (relative thermodynamic stabilities and chemical shift values).
O group and those lacking it. We performed DFT calculations to gain some insight into the structures of the inclusion complexes and support the experimental findings (relative thermodynamic stabilities and chemical shift values).
Sierra, A. F.; Aragay, G.; Gomila, R. M.; Ballester, P.
Org. Chem. Front. 2023, (10), 996-1002
DOI:
10.1039/d2qo01952c

Let's create a brighter future
Join our team to work with renowned researchers, tackle groundbreaking
projects and contribute to meaningful scientific advancements




















