H-Bonded Counterion-Directed Enantioselective Au(I) Catalysis
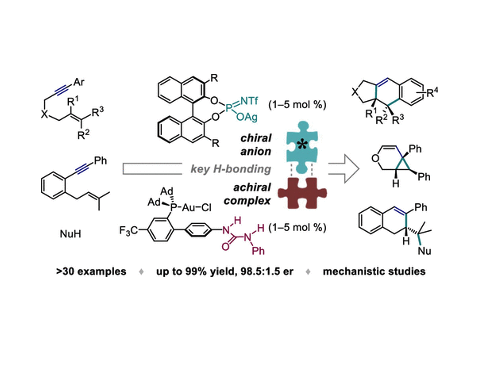
A new strategy for enantioselective transition-metal catalysis is presented, wherein a H-bond donor placed on the ligand of a cationic complex allows precise positioning of the chiral counteranion responsible for asymmetric induction. The successful implementation of this paradigm is demonstrated in 5-exo-dig and 6-endo-dig cyclizations of 1,6-enynes, combining an achiral phosphinourea Au(I) chloride complex with a BINOL-derived phosphoramidate Ag(I) salt and thus allowing the first general use of chiral anions in Au(I)-catalyzed reactions of challenging alkyne substrates. Experiments with modified complexes and anions, 1H NMR titrations, kinetic data, and studies of solvent and nonlinear effects substantiate the key H-bonding interaction at the heart of the catalytic system. This conceptually novel approach, which lies at the intersection of metal catalysis, H-bond organocatalysis, and asymmetric counterion-directed catalysis, provides a blueprint for the development of supramolecularly assembled chiral ligands for metal complexes.
Franchino, A.; Martí, A.; Echavarren, A. M.
J. Am. Chem. Soc. 2022
DOI:
10.1021/jacs.1c11978

Let's create a brighter future
Join our team to work with renowned researchers, tackle groundbreaking
projects and contribute to meaningful scientific advancements




















