Dichloromethane as C1 Synthon for the Photoredox Catalytic Cyclopropanation of Aromatic Olefins
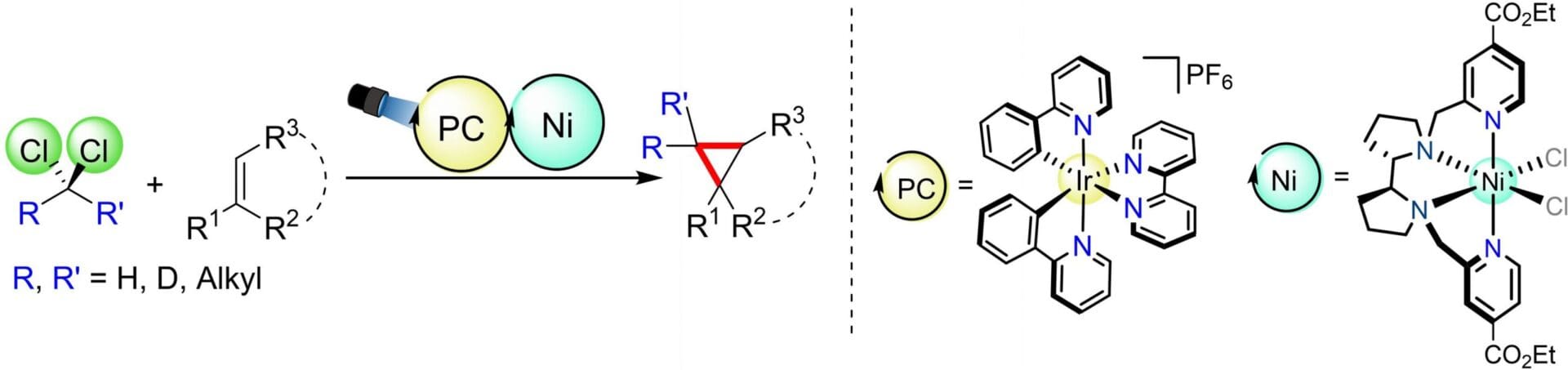
Dichloromethane, as a readily available and inexpensive C-1 synthon is proposed as a powerful building block for cyclopropanation of alkenes under mild conditions. Herein, we report a highly efficient and versatile dual photoredox system, involving a nickel aminopyridine coordination complex and a photocatalyst, for the cyclopropanation of aromatic olefins using dichloromethane, under visible-light irradiation. The cyclopropanation protocol has been successfully applied at gram scale. Mechanistic studies suggest a Ni(II) pyridyl radical complex as the key intermediate for the homolytic cleavage of the C-sp3-Cl bond, generating a chloromethyl radical that is captured by the olefin coupling partner. Our findings also highlight the versatility of this methodology. By directing the radical/polar crossover process, we were able to selectively drive the reaction towards either the formation of cyclopropyl derivatives or the corresponding non-cyclic alkyl chloride products. The methodology also successfully apply to geminal dichloroalkanes, including the formation of spiro[2,2] compounds. Moreover, our methodology extends to the synthesis of deuterium-labelled cyclopropanes, demonstrating its utility in isotopic labelling and broadening its applicability in chemical synthesis and drug development.
Aragon, J.; Sun, S.; Fernandez, S.; Lloret-Fillol, J.
Angew. Chem.-Int. Edit. 2024,
DOI:
10.1002/anie.202405580

Let's create a brighter future
Join our team to work with renowned researchers, tackle groundbreaking
projects and contribute to meaningful scientific advancements



















