Binding Studies of a Luminescent Pt(II) Organometallic Calix[4]pyrrole with Halide Salts
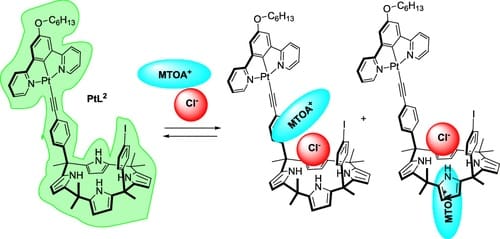
We report the synthesis of a luminescent Pt(II) complex, PtL 2 , based on a two-wall aryl-extended calix[4]pyrrole (C[4]P). We characterize its binding properties as a receptor of methyl trioctyl ammonium chloride (MTOACl) in a dichloromethane solution. To this end, we performed 1H NMR and UV-vis spectroscopic titrations. The singular luminescent properties of PtL 2 allowed the use of highly sensitive emission spectroscopy to monitor the binding. The binding affinity of PtL 2 for MTOACl is 2-fold smaller than that of the C[4]P precursor. This difference is attributed to dissimilar anion-pi interactions and binding geometries in the ion-paired complexes of the two receptors, (Cl subset of PtL 2 /C[4]P)@MTOA. Isothermal titration calorimetry (ITC) experiments revealed subtle differences in enthalpy and entropy. The entropic term, although negative, was very small, suggesting the relevance of the dissociation/association processes of the salt and the ion-paired complexes. We investigated the binding selectivity of PtL 2 toward a series of halides in acetone solution favoring ion-pair dissociation. PtL 2 binds chloride selectively and with a high affinity, producing mainly an anionic complex.
Lázaro, A.; Valencia, E.; Ballester, P.; Rodríguez, L.
ACS Omega 2025, 10 (8), 8665-8674
DOI:
10.1021/acsomega.4c11558
Associated projects:
-
COMPOFUN
The overarching aim of this project is the synthesis of molecular and supramolecular containers / receptors with polar interiors derived from the use of aryl- and super-aryl- extended calix[4]pyrroles, and the evaluation of their applications in different areas ranging from fundamental studies of physical organic chemistry, the development of sensor devices, molecular recognition, supramolecular catalysis and the transport of molecules across lipid membranes
See more

Let's create a brighter future
Join our team to work with renowned researchers, tackle groundbreaking
projects and contribute to meaningful scientific advancements






















