A paper by Muñiz group on the cover of Angewandte Chemie International Edition
 Structurally Defined Molecular Hypervalent Iodine Catalysts for Intermolecular Enantioselective Reactions
Structurally Defined Molecular Hypervalent Iodine Catalysts for Intermolecular Enantioselective Reactions
S. Haubenreisser, T. H. Wöste, C. Martínez, K. Ishihara, K. Muñiz
Angew. Chem. Int. Ed., 2016, 55, 413-417
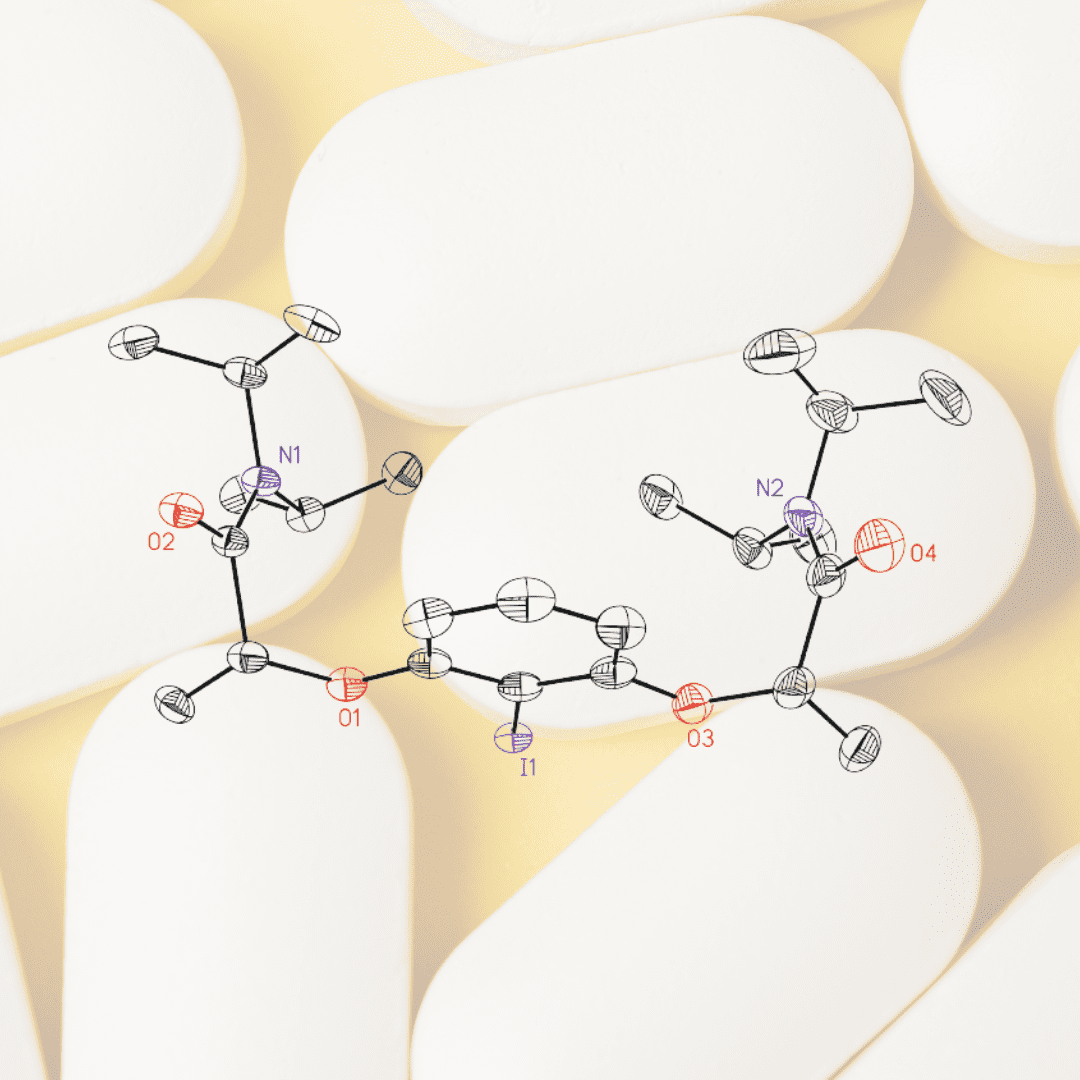
Molecular structures of the most prominent chiral non-racemic hypervalent iodine(III) reagents to date have been elucidated for the first time. The formation of a chirally induced supramolecular scaffold based on a selective hydrogen-bonding arrangement provides an explanation for the consistently high asymmetric induction with these reagents. As an exploratory example, their scope as chiral catalysts was extended to the enantioselective dioxygenation of alkenes. A series of terminal styrenes are converted into the corresponding vicinal diacetoxylation products under mild conditions and provide the proof of principle for a truly intermolecular asymmetric alkene oxidation under iodine(I/III) catalysis.
Related news

Let's create a brighter future
Join our team to work with renowned researchers, tackle groundbreaking
projects and contribute to meaningful scientific advancements






 27-05-2022
27-05-2022