Englerin analogues with anti-cancer activity
29th April 2016 – The research group of Prof. Echavarren has designed and synthetized a series of analogues of (-)-Englerin A that are highly selective and effective in the growth-inhibition of renal cancer cells.

Molecular structure of (-)-englerin A
The molecule (-)-Englerin A was isolated in 2009 from the bark of the east African plant Phyllanthus engleri by scientists at the US National Cancer Institute, and has received a lot of attention because of its ability to kill kidney cancer cells selectively. Kidney cancer is often treated surgically and responds poorly to existing drugs, which can have serious side effects. So, new drugs based on (-)-Englerin A could help improve outcomes for patients. Several total syntheses have been reported to date and two distinct mechanisms for Englerin A’s anticancer activity have been proposed.
In 2010 the research group of Prof. Echavarren achieved the total synthesis of (-)-Englerin A starting from inexpensive geraniol and using as the key transformation a gold-catalyzed cycloaddition invented by his group. The synthesis was efficient and easily scalable and, what is more important, the intermediate obtained after the gold-catalyzed cycloaddition opened the way for the synthesis of new and useful derivatives.
Taking the previous total synthesis of the natural product as starting point, Echavarren’s group has synthetized a set of analogues with great structural diversity. An important point to note is that the original synthesis has been scaled-up to the multigram scale. “We have developed potent anticancer compounds inspired on the structure of Englerin as a final result of curiosity-driven research in gold chemistry” -says Prof. Echavarren.
The biological study of the first series revealed that the derivatives with a double bond between C4 and C5 were more active against renal cancer and worthy of further exploration. Then, a second library of compounds maintaining the unsaturation between C4 and C5 was prepared. The study of the new series of compounds revealed and interesting selectivity profile and very good growth inhibition potency in some of the renal cancer cell lines. The structure-activity relationship findings of this study open promising ways in the fight against renal cancer and are being used in the design of more potent and bioavailable drug candidates against other cancer types.
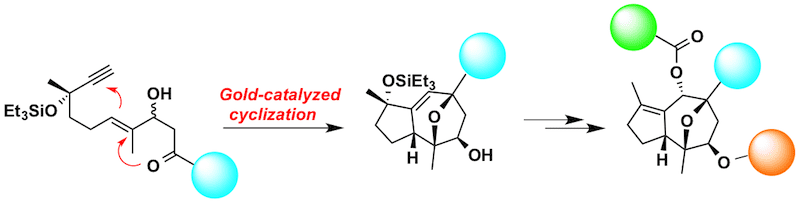
General synthesis of (-)-englerin derivatives
The program initiated by basic research in the new field of gold catalysis led to the total synthesis of a biologically relevant molecule, opening the doorway to a practical solution for the synthesis of structurally simplified analogues, which were developed in the CSOL (Catalyst Selection and Optimization Laboratory) unit at ICIQ. This Technology Development Unit is oriented the valorization of research results developed at ICIQ in order to bridge the gap between research results and their application in industry. The biological studies have been carried out in the US National Cancer Institute (National Institute of Health, USA) by Prof. Beutler and collaborators.
A PCT patent application has been filed for these results jointly with the National Institute of Health (USA).
Ref:
Synthesis and Biological Evaluation of New (-)-Englerin Analogues
López-Suárez, L. Riesgo, F. Bravo, T. T. Ransom, J. A. Beutler, A. M. Echavarren
ChemMedChem, 2016, DOI: 10.1002/cmdc.201600040
Media:
Related news

Let's create a brighter future
Join our team to work with renowned researchers, tackle groundbreaking
projects and contribute to meaningful scientific advancements






 06-03-2025
06-03-2025 



















