The first π-coordination-catalyzed nucleophilic fluorination of unactivated aryl halides has been demonstrated. Chlorobenzene reacts with alkali metal fluorides (CsF, KF) in the presence of a Cp*Ru catalyst at 120–180 °C to give fluorobenzene.
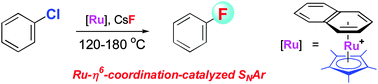
The first π-coordination-catalyzed nucleophilic fluorination of unactivated aryl halides has been demonstrated. Chlorobenzene reacts with alkali metal fluorides (CsF, KF) in the presence of a Cp*Ru catalyst at 120–180 °C to give fluorobenzene.
