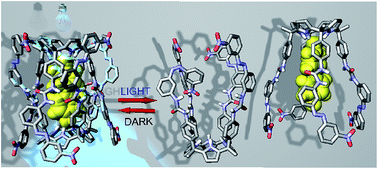
A tetraurea aryl extended calix[4]pyrrole with four appended azobenzene groups dimerizes by encapsulating a bis-N-oxide acting as a template. The assembly can be detected by 1H NMR spectroscopy only when all eight azobenzene units are in their trans forms. The light-induced trans-to-cis-isomerization of a single azobenzene moiety within the assembly triggers capsular disintegration, probably through a disassembly process. The reassembly of the encapsulation complex is achieved by cis-to-trans relaxation of the azobenzene photoswitches in the dark.