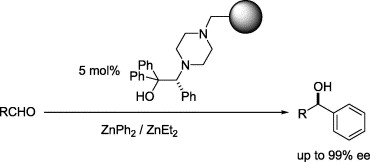
A family of polystyrene-supported amino alcohols, characterized by a high catalytic activity in alkyl transfer from zinc to formyl groups has been successfully tested in the enantioselective addition of phenyl zinc reagents to aldehydes to afford chiral diarylmethanols. Enantioselectivities higher than 90% (mean ee 90.5%; eight examples) are recorded with aromatic aldehydes in what represents the first successful use of heterogeneous, polymeric reagents for enantiocontrol in the phenylation of aldehydes.