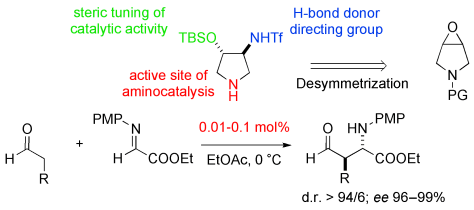
A family of enantiopure 4-oxy-substituted 3-aminopyrrolidines arising from the enantioselective ring-opening of meso-3-pyrroline oxide have been developed as catalysts for the asymmetric, anti-selective Mannich reaction (see scheme; PMP=p-methoxyphenyl; PG=protecting group). Very high catalytic activity (down to 0.01 mol % loading) and stereoselectivity have been recorded.