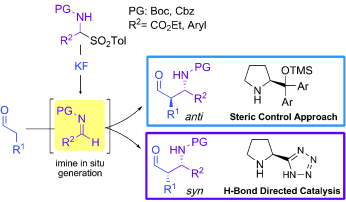
A simple and convenient method for the direct, aminocatalytic, and highly enantioselective Mannich reactions of aldehydes with in situ generated N-carbamoyl imines has been developed. Both α-imino esters and aromatic imines serve as suitable electrophilic components. Moreover, the judicious selection of commercially available secondary amine catalysts allows selective access to the desired stereoisomer of the N-tert-butoxycarbonyl (Boc) or N-carbobenzyloxy (Cbz) Mannich adducts, with high control over the syn or anti relative configuration and almost perfect enantioselectivity. Besides the possibility to fully control the stereochemistry of the Mannich reaction, the main advantage of this method lies in the operational simplicity; the highly reactive N-carbamate-protected imines are generated in situ from stable and easily handled α-amido sulfones.